Andrea Corbo, Cecilia Luci, and Giovanni Merone discuss the findings from their study evaluating dosage, patient compliance, degree of satisfaction, and treatment protocol for equine collagen injections to treat the signs of ageing
In recent years there has been a growing interest in rejuvenating therapies for cosmetic medicine. These are based on both physical methods, such as radiofrequency, laser and pulsed light, and biochemical methods through injectables. Several injectables are available for this purpose, comprised of non-crosslinked hyaluronic acid, polynucleotides, vitamins, organic silicon, and others.
Product characterization and rational
Equine collagen type I has been used in dermatology as well as plastic and vascular surgery for wound healing and skin ulcer therapy for at least three decades1–7. The most common product existing is based on lyophilized collagen tablets, generally placed directly into the wounds to stimulate tissue regeneration and accelerate healing. The equine collagen is obtained from the Achille’s tendon of a horse. During the production process, the collagen molecule does not undergo chemical changes in its structure, such as protein denaturation or cross-linking. The only chemical manipulation is separation from N and C–terminal amino acid sequences of the collagen triple helix, to reduce the risk of allergy. Equine collagen has documented effects on fibroblast mitosis, tissue regeneration and new collagen biosynthesis stimulation1–7.
ust be injected intradermally.
The supposed mechanism of action is based primarily on the presence of tripeptides, in particular, Pro-Hyp-Gly derived from collagen hydrolysis. In fact, an hydrolysis of peptidic bonds could occur both in vivo, after product injection in the receiving tissue, and in vitro, in the same vial after product reconstitution. Once in thesolution, equine collagen undergoes spontaneous hydrolysis, producing tripeptides and collagenous amino acids. This guarantees the total absence of any risk of immunogenicity because the molecules capable of eliciting an immune response must be larger than 10 kDa, while the molecules in the solution are 3–6 kDa at a maximum (tripeptides)8.
Tripeptides are known as biological messengers and act similar to growth factors; interacting with transmembrane receptors, they activate anabolic pathways and stimulate the fibroblast cell cycle. The Pro-Hyp-Gly tripeptide works as a competitive inhibitor of matrix metalloproteinases (MMP), which is responsible for the enzymatic degradation of dermal collagen (collagenase)9–12.

Some in vitro studies have shown that younger fibroblast, i.e., resulting from cell mitosis, synthesise principally type III collagen, while older fibroblasts mostly synthesise type I. It is known that the collagen I/collagen III ratio increases during skin senescence13.
Materials and method
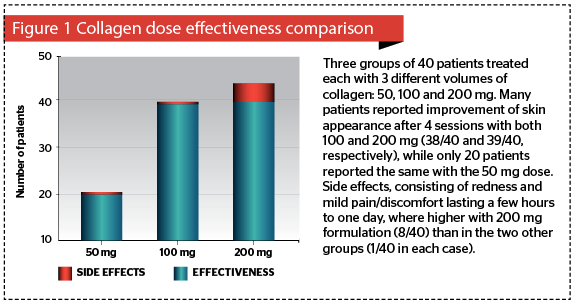
To find the appropriate dosage, three different quantities of equine collagen were tested on 120 patients, aged between 35 and 65 years, divided into three groups of 40 subjects, each group was treated with a different dose: 50, 100 and 200 mg of micronized sterile powder, prepared with 0.5 ml of 2% lidocaine solution plus 4.5 ml of saline. The resulting suspension was injected intradermally using the mesotherapy point-by-point technique, intradermal papules were spaced 2 cm along the face, neck, décolleté and hands (about 0.1 ml per injection). The four individual treatments were scheduled every 2 weeks across 8 weeks. Photographic documentation was collected before and after the treatment protocol. All side-effects were recorded. Patients were asked to complete a self-assessment questionnaire, to gain their opinion on skin texture, brightness, hydration, and firmness.
The effectiveness on skin improvement was evaluated by both self-assessment and operator’s evaluation, as comparable in both formulations of 100 and 200 mg, while effectiveness was lower with the 50 mg. Side-effects, mainly redness, warmth and transient pain/discomfort, were higher in terms of frequency in the 200 mg formulation. The 100 mg formulation of equine collagen was chosen finally for further treatments since it demonstrated the best ratio between efficacy and side-effects. (Figure 1)
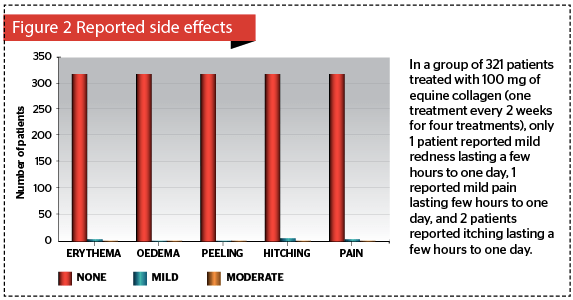
A second group of 321 patients were then treated with 100 mg equine collagen, according to the protocol described above (100 mg of powder suspended in 4.5 ml of saline and 0.5 ml of lidocaine 2%, for total solution volume of 5 ml), for a full treatment to their face, neck, decollettè, and hands (0.1 ml every 2 cm, one session every 2 weeks for four sessions). Photographic documentation and self-assessment questionnaires were collected as in the previous group.
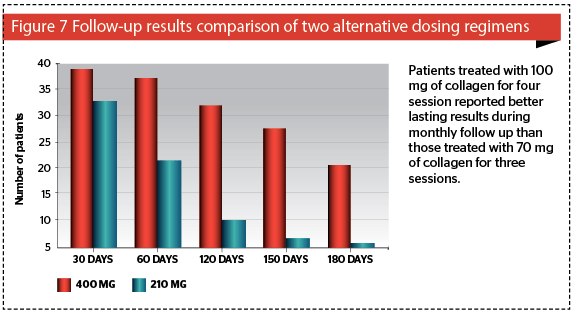
In the last phase of the authors’ study, they tested the best protocol for result maintenance, performing monthly check-ups until 6 months on a fourth group of 80 patients divided into 2 sub-groups of 40 patients (A and B) treated with two different doses of collagen. Group A was treated with 70 mg of collagen in each session for 3 sessions every 2 weeks for a total dose of 210 mg; while group B was treated with 100 mg of collagen in each session for a total of 4 sessions every 2 weeks for a total of 400 mg. Patients were monitored up to 6 months, with clinical evaluation and a self-assessment questionnaire.
Results
All enrolled patients completed the study, with no drop-off occurring.
In the second group of 321 patients treated with 100 mg of equine collagen over four sessions, data collected from the self-assessment questionnaire is outlined below.
Patient evaluations revealed a significant improvement of texture, consistency, brightness, and skin hydration in more than 95% of cases (Figure 3).
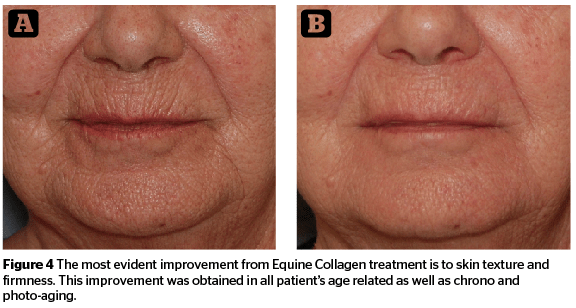
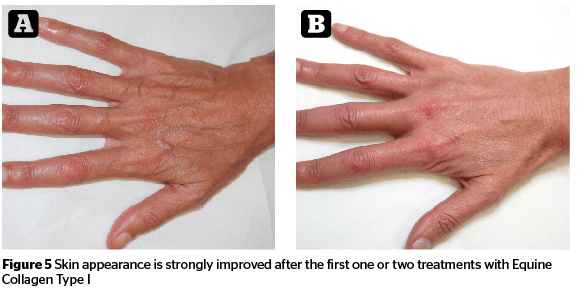
Photographic documentation showed improvement in chrono- and photo-aging (Figures 4 and 5).
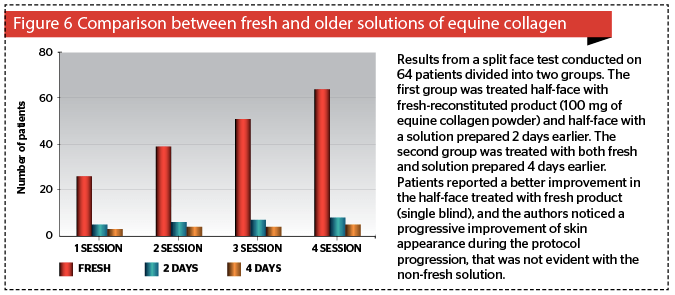
In the split face test (the third group studied) the authors found a significant reduction in effectiveness when non-fresh product was used. Product that had been made days in advance instigated less of a treatment response. Areas in which the authors used solution prepared 4 days earlier showed a very poor response, while the preparation stored for two days before treatment responded a little better. Only the areas treated with freshly formulated suspension showed the expected results, as revealed by patient evaluation (Figure 6). The reported data confirmed clinical judgment.
Discussion
In the field of cosmetic medicine, we are observing an increasing request for skin appearance improvement, and a more rejuvenating-like effect from treatments rather than only corrective or volume shaping outcomes. For this reason, a lot of injectable products are currently available on the market for rejuvenating purposes. In particular, non-crosslinked hyaluronic acid is widely used. Hyaluronic acid for skin rejuvenation predominantly effects dermal hydration, associated with the ROS scavenger effect, but it is not free from doubt14,15.
On the other side, in fact, it seems to activate metalloproteinase gene expression, leading to an increase in dermal matrix degradation14,15. For this reason, it is recommended to associate the use of other products for bio-stimulation that can increase dermal collagen production and shut down metalloproteinase. The main limit of classical bio-rejuvenation/bio-stimulation injective procedures (polynucleotides, vitamins) is the residual anabolic capacity of old fibroblasts. Therefore, the novel use of equine collagen, which is proven to stimulate fibroblast mitosis and tissue regeneration, should be explored. Younger fibroblasts resulting from mitosis should have greater anabolic capacity and should be committed to producing a greater amount of collagen type III than the older fibroblasts. Finally, the equine collagen has no allergic reaction reported in more than 3 decades of use, showing a high degree of safety, which is essential for a cosmetic injection procedure.