Androgen-dependent female hair loss is a disease affecting 40–50% of all women1. Its onset may occur at any age between puberty and the menopause, and it can be triggered by a number of different mechanisms. Essentially, it is supported by a lack of oestrogen and takes the form of progressive miniaturisation of the single hairs. In this study, the authors proposed to investigate the therapeutic effect of a product based on polydeoxyribonucleotides in women with hormone-dependent hair loss, administered by means of intradermal infiltrations carried out according to standard timing for a period of 4 months. The results were assessed by means of photographic documentation, pull tests, trichograms, wash tests and dermatoscopy for counting the average number of hairs and the diameters of the hair shafts, at time 0 and after 30, 60 and 120 days. The results achieved were satisfactory for all the patients and no side-effects were observed. The authors found that all patients were generally satisfied, and this feeling increased progressively alongside the objective results. The pull tests normalised progressively and the trichograms recorded gradual recovery of the anagen-telogen ratio, with an increase in the number of hairs in the anagen phase. The number of hairs falling out decreased (wash test) and, consequently, the hair became thicker and the structural size of the shafts increased (dermatoscopy). To conclude, the authors believe that the results achieved confirm the trophic stimulus exercised by the polynucleotides on the hair follicles of the scalp for female hair loss.
Female hormonal hair loss (female androgenetic alopecia; FAGA) affects approximately 40–50% of all women1, often causing significant psychological repercussions on top of the damage in functional terms and of appearance. The onset of female hair loss can occur at any age, from puberty right through to old age; indeed, whenever changes in the oestrogen–progesterone balance take place2.
Possible triggering factors include the start or suspension of contraceptive therapy, pre-partum or post‑partum modifications, pregnancy, stressful events or dietary deficiencies.
Female hormonal hair loss
Thinning of the hair can occur in a number of ways:
- In the prevailing pattern (female pattern) a reduction in the concentration of the hair is observed in a triangular-shaped frontal area behind a persistent fringe (Christmas tree pattern)
- The thinning may affect the vertex area (Ludwig Pattern) in accordance with Types I, II or III, depending on how severe the hair loss is
- Very rarely, the male model (androgen-dependent Hamilton pattern) is observed, with a receding hair line on the forehead and thinning on the vertex3.
In non-androgen-dependent female hormonal hair loss, observation of the skin of the scalp reveals areas featuring thinning of the hair owing to the presence of empty follicles as a result of a lack of synchronism between a hair falling out and the appearance of a newly formed hair to take its place. At least in the initial stages, replacement takes place only after a fairly long period of time.
The consequence of this is a smaller number of hairs in the growth (anagen) phase, which lasts less than usual (shorter and finer hair) and an increasingly high number of hairs entering the telogen phase, which also lasts for a longer period of time (i.e. alopecia)4, 5. Actual baldness rarely occurs, but almost always, areas of alopecia arranged in mosaic-like fashion are observed, in which the number of hairs is smaller and there is a lower proportion featuring a normal structure mixed with fine hairs (vellus)6, 7.
The cause of female hormonal hair loss is, essentially, the absence of oestrogens (oestrone) or their failure to work, with progressive miniaturisation of the hairs (papilla, matrix and shaft), so that only fine and thinned hair grows all over the scalp (mainly on the vertex and in the frontal area), with normal menstruation and fertility, no excess of circulating androgens, and the absence of indices of telogen effluvium. Rather than alopecia, the clinical appearance is that of hypotrichia.
Polynucleotides
The possible treatment strategies are aimed, on the one hand, at limiting the action of the hormones on the hair follicles and on the other, at providing effective stimulation of cell proliferation and of growth of the hair adnexum.
It is well known that polynucleotides (PN) have the trophic effect of stimulating cell proliferation on a number of different cell core types, including fibroblasts and adipocytes8–10. PN are fractions of DNA with a low molecular weight, of natural origin. They are obtained by means of a process of extraction from trout sperm using high-temperature purification and sterilisation, without the interaction of peptides or pharmacologically-active proteins. This compound contains a mixture of deoxyribonucleotide polymers with chain lengths ranging between 50 and 2000 bp, and can also be the source of purine and pyrimidine deoxynucleosides/deoxyribonucleotides and bases.
The trophic effect of PNs appears to be mediated by stimulation of the A2 purinergic receptors (adenosine receptors), thus promoting the metabolic activity of the cells. Furthermore, nucleotides promote the metabolic ‘salvage’ pathway, rather than the traditional ‘ex novo’ synthesis starting from amino acids8, 11. Since purinergic receptors are present also in the hair follicles, and trophic stimulation appears to be useful in the event of female hormonal hair loss for facilitating the hair growth phase12–14, the authors assessed the effects of PN administered by intradermal infiltration.
Materials and methods
Patient selection
The authors selected 20 female patients aged between 25 and 65 years, all suffering from non-androgen-dependent female hormonal alopecia (i.e. female hormonal hair loss). The exclusion criteria were current or previous systemic diseases capable of increasing hair loss (low serum iron levels, fever, endocrine diseases, liver diseases), medical history of current or recent slimming diets, current or recent conditions of psychophysical stress, and pharmacological systemic treatments and/or contraceptive pills. Of these 20 patients, five had already undergone treatment with local stimulating lotions (not otherwise specified) and supplements. The others had not received any previous treatment.
With regard to their case histories, none of the patients reported any current or earlier systemic diseases able to accentuate their hair loss (e.g. low serum iron levels, febrile diseases, endocrine diseases, liver diseases) and none reported having followed slimming diets. No current or recent conditions of psychophysical stress were reported. None of the patients examined were receiving pharmacological treatment or taking contraceptive pills. Two patients reported that they were mild smokers, with a daily average of 6–8 cigarettes.
All patients appeared to be in good conditions of health, with the presence of progressive thinning of the hair adnexa on their scalps. All patients provided informed consent.
Methods
PN were administered using pre-filled syringes (Plinest® Fast 7.5 mg/ml; Mastelli Ltd, Sanremo, Italy) by means of intradermal infiltration, using the serial puncture technique and injecting 0.2 ml of product for each puncture at a distance of 3–5 cm from one another. The product was distributed uniformly throughout the area affected by alopecia by administering one whole syringe (1.3 ml) per patient at each infiltration session.
The treatment protocol called for one infiltration session per week for the first 4 weeks, followed by one every 2 weeks for 3 months. Accordingly, the treatment lasted for an overall period of four months, with a total of 10 infiltration sessions.
The patients were advised to wash their hair three times per week using a mild shampoo.
Clinical assessments were carried out at the start of the treatment (T0), after 1 month (T30), after 2 months (T60), and at the end of the 4-month study period (T120).
The examinations included clinical observation, a number of different instrumental tests, and the patients’ own subjective considerations. The instrumental tests were:
- Photographic documentation (global photo)
- Pull test15, 16
- Trichogram15, 16
- Count of fallen hairs (wash-test carried out by the patient after washing her hair)15, 16
- Dermatoscopic examination, for assessing the physical parameters and density of the hair3 performed by DermLite Foto system17.
Product
The product is a gel comprising highly purified PN of natural origin (Plinest® Fast, 7.5 mg/ml). It is supplied in pre-filled glass syringes, each containing 1.3 ml of high molecular weight, sterile, and apyrogenic PN. It is a biocompatible class III medical device (CE 0373) of natural origin and allergy testing is not necessary. The product has undergone many in vitro and in vivo tests18,19, showing the absence of local and systemic toxicity, in accordance with UNI standards for medical devices (cytotoxicity by direct contact, allergic sensitisation, Ames mutagenicity test, intracutaneous reactivity, irritation test). In clinical use, high tolerability and the total absence of serious side‑effects have been reported. The PNs were kindly supplied free of charge (Plinest® Fast).
Results
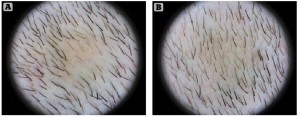
Figure 5 Videodermoscopy: average number of hairs in a standard surface pre- and post-treatment (T0 vs T120). (A) T0, 105 hairs/cm2; (B) T120, 118 hairs/cm2
The treatment was carried out in accordance with the protocol on all the patients enrolled in the study. No local or systemic side-effects requiring suspension of the treatment were observed.
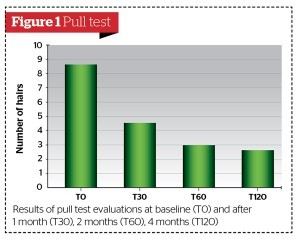
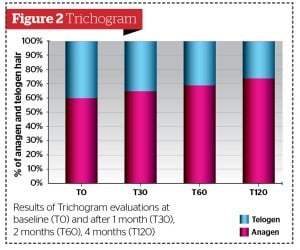
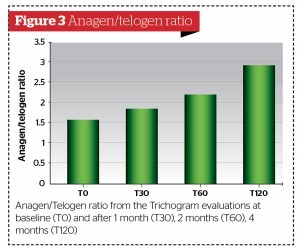
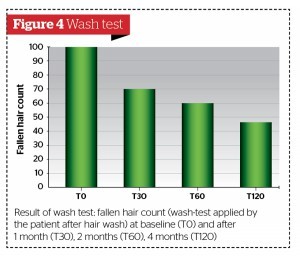
The results of the treatment were, overall, highly satisfactory, as shown in the tables provided, with a marked reduction in hair loss (Figures 1–4) alongside an increase in the number of and diameter of the hair shafts (Figures 5 and 6). Furthermore, the trichogram (Figure 2) values and the pull test (Figure 1) values of the hair returned to a relatively ‘normal’ state in all patients treated.
As far as compliance is concerned, the patients accepted the treatment willingly, also considering the clearly noticeable clinical results (Figure 7), as well as the objective data acquired.
Discussion
Thellung et al11 and Sini et al8 demonstrated that PN enhanced the proliferation of human fibroblasts in vitro. Belletti et al studied the effect of PN on UVB-damaged fibroblasts. The study showed that PN increased the rate of repair of UVB-damaged dermal fibroblasts20 and appeared to promote the proliferation of human pre‑adipocytes9. A recent study evaluated the effect of PN and hyaluronic acid (HA) on cultured human fibroblasts by analysing cell growth10. The results confirmed the activity of PN on human fibroblasts, while no significant stimulation was obtained with HA alone, demonstrating the trophometabolic action of polynucleotides in different cell lineage 10.
PN are considered important bio-stimulating agents in aesthetic medicine owing to their specific action on fibroblasts, as shown by a number of clinical studies investigating their use21–24.
With regard to the scalp, tests were conducted in the past on patchy alopecia25, and an initial test was more recently conducted for the treatment of androgenic alopecia with PN (7.5 mg/ml)26. This study was conducted with the same protocol of treatment used in above trial. It showed a positive efficacy that was evaluated by pre and post-treatment digital pictures and by physician assessments; in 72% of the patients the physician noted a clinical improvement of female androgenetic alopecia with no side effects and high patient compliance.
For these reasons, the authors also started to use them in trichology in order to investigate the possibility of stimulating trophism of hair follicles in poor condition.
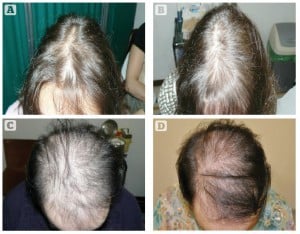
Figure 7 Clinical results before and after 4 months. Patient. 1 (A) before and (B) after treatment. Patient 2 (C) before and
(D) 4 months after treatment
It is well known that fibroblast growth factor (FGF) is extensively involved in the development of hair follicles, and is considered an important mediator in their metabolism and differentiation. Immunohistochemical studies found A2 receptors (adenosine receptors) in the dermal papilla cells of hair follicles27. It has been shown12 that incubation of the hair adnexa with 25 μm of adenosine significantly increased the concentration of intracellular AmpC in the dermal papillae. To be precise, the production of AmpC appeared to depend specifically on the interaction of adenosine with A2 receptors28–30. It would therefore seem reasonable to assume that AmpC or similar substances are crucial for the positive control of FGF-7, which regulates the development of the hair follicles. Specifically, studies were conducted on cultures of hair follicles in the anagen phase incubated with FGF-712. A more significant growth in length was observed by comparison with control cultures after only 6 days, as well as a longer duration of the anagen phase in the context of increased proliferation and differentiation of the follicle cells12.
In the treatment of androgenic alopecia Minoxidil is the only approved topical treatment, but the exact mechanism by which Minoxidil induces hair growth is still unknown. Some authors suggest that Minoxidil improves the release of adenosine, which in turn acts on specific adenosine receptors of hair follicles releasing vascular endothelial growth factor27.
This mechanism of action is particularly interesting as, since they are DNA fractions, PN gradually release adenosine during catabolism. Thus, the stimulation of dermal papilla cells could be the result of the action performed by adenosinic nucleotides on A2 purinergic receptors.
These considerations tend to support the effectiveness of PN the authors observed in the clinical assessment based on PN administration. These proved to be molecules of the very first choice when significant stimulation of the cells of the dermal papillae is required in order to support the trophometabolic activity, thus causing a substantial improvement in cases of female hormonal hair loss.
Conclusions
The results of this clinical evaluation confirm that trophic stimulation performed by PN in the scalp promoted the growth of hair in cases of female hormonal hair loss.
This clinical investigation also confirmed the safety of the medical device and its good tolerability.
According to the protocol presented in the study, polynucleotides for scalp injections are an effective, safe, and non invasive procedure for patients. Other surgeons can easily repeat the treatment. Despite the clear limits of the authors’ study owing to the reduced size of the sample, as far as they are aware, these are the first data available on the efficacy of polynucleotides evaluated on the basis of different objective criteria (trichogram, hair count, hair density, pull test). They show that the treatment with polynucleotides is effective in improving hair regeneration and in counteracting the female hormonal hair loss. In the light of these results it could be interesting to evaluate the action of polynucleotides on hair regeneration following different pattern of hair loss, such as alopecia areata and male androgenetic alopecia.
Polynucleotides could be also useful as adjuvant post surgery treatment after hair transplant surgery. All the above mentioned clinical conditions could be the objects of further interesting studies and trials.